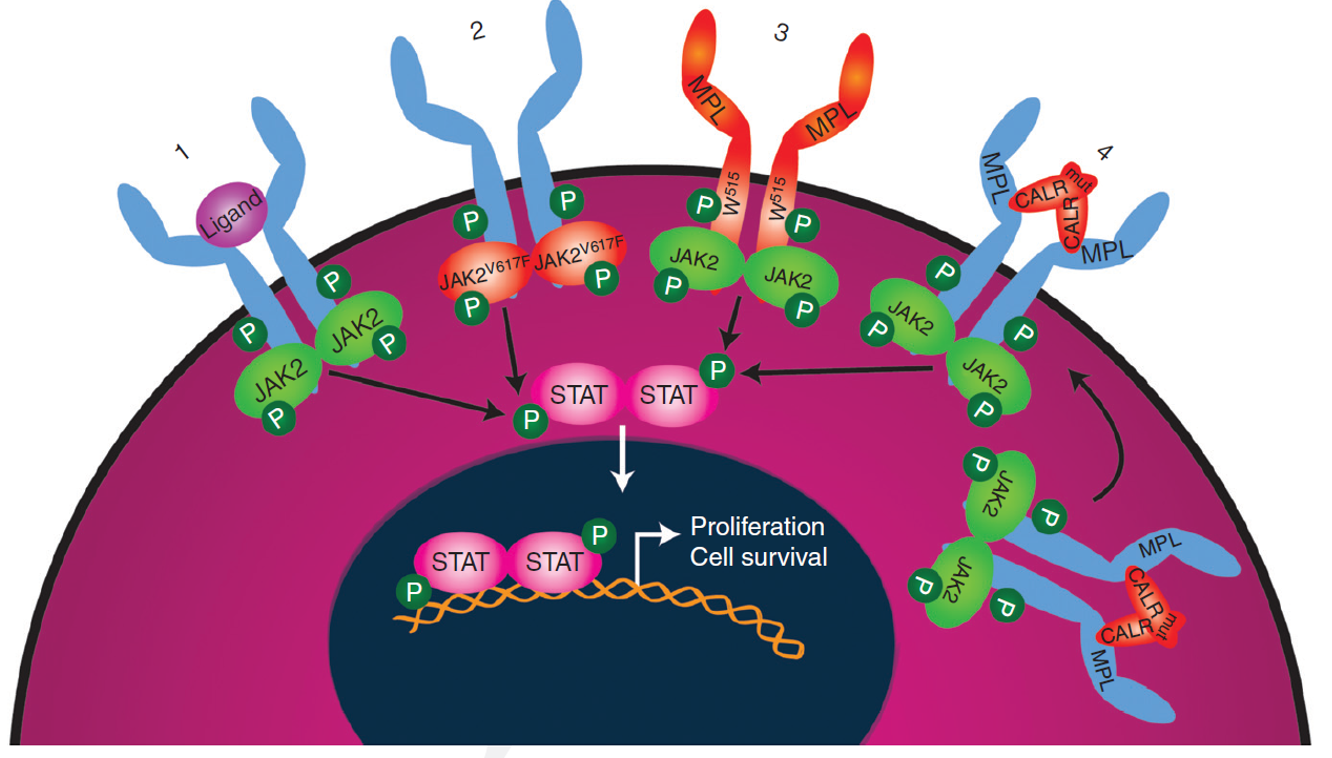
Building on our work elucidating the mechanism by which mutant CALR induces MPN (Elf et al., Cancer Discovery 2016, Elf et al., Blood 2018), we are now focused on identifying unique molecular dependencies of mutant CALR MPN stem cells and on developing novel therapeutic approaches to inhibiting mutant CALR (including immunological approaches). We are using molecular and cellular biology approaches in addition to mouse models, functional genomics, proteomics and single cell approaches to address these questions.
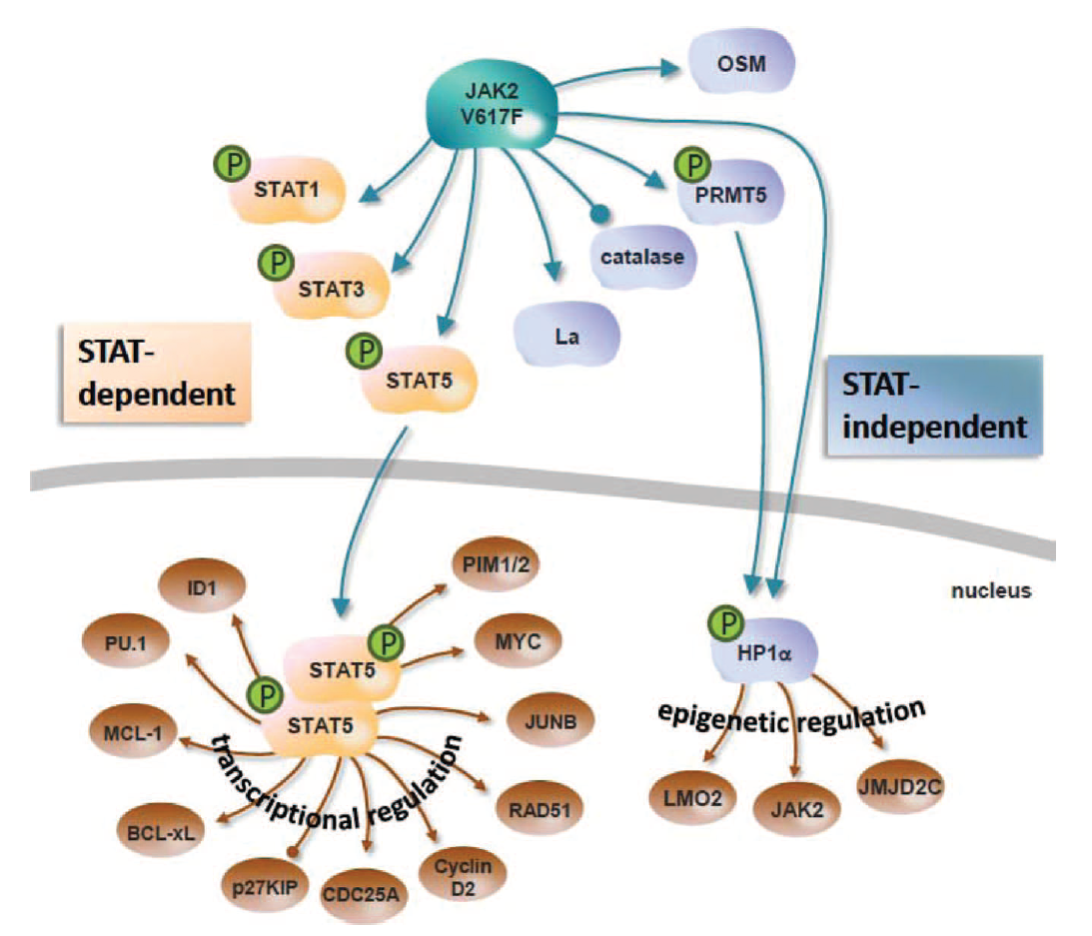
The JAK2V617F mutation is central to MPN pathogenesis and is a common cause of clonal hematopoiesis of indeterminate potential (CHIP). Our lab has several projects involving both human and mouse to understand the impact of the JAK2V617F mutation on hematopoiesis and to identify novel approaches to preferentially therapeutically target JAK2V617F MPN stem cells.
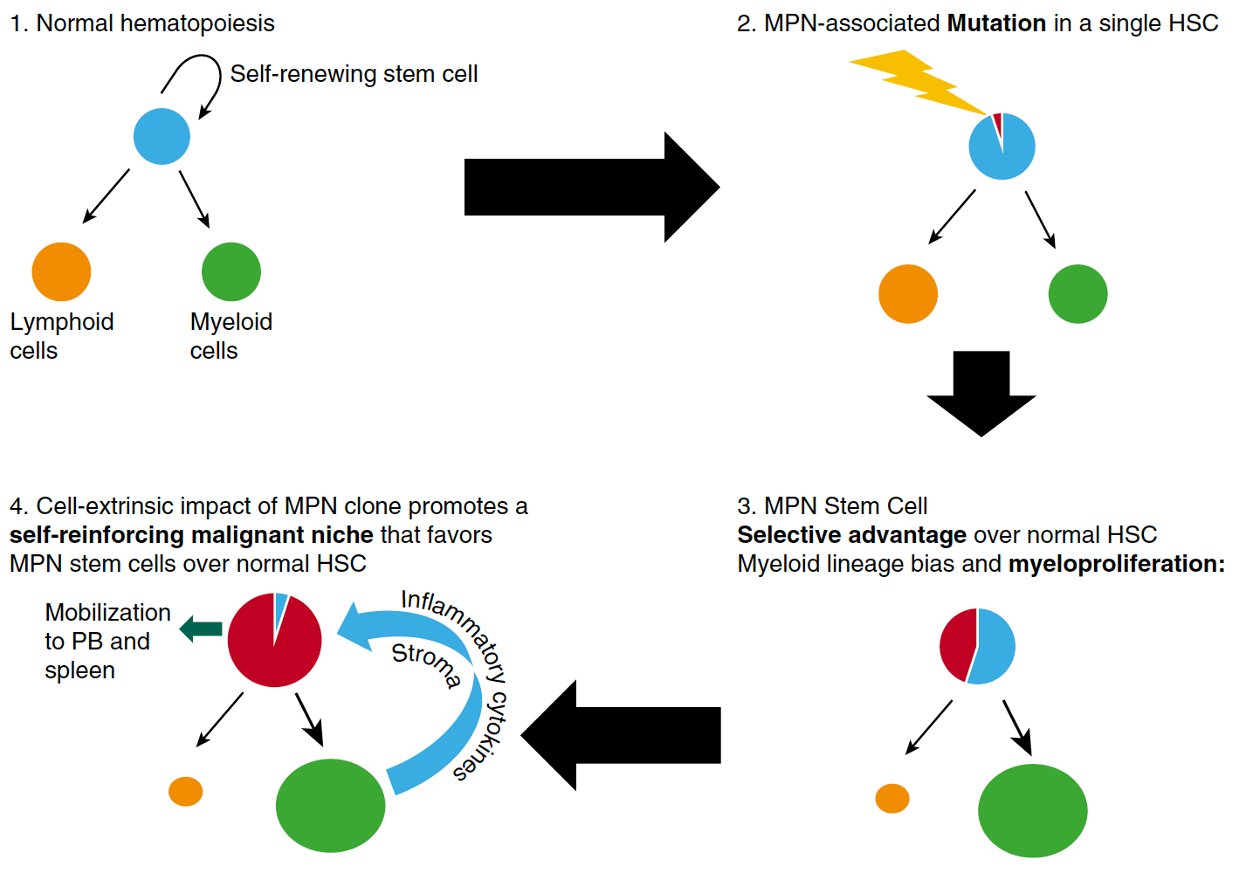
A key question in MPN is why some patients have stable clinical disease over decades whereas other patients can develop disease progression in a relatively short period of time. Using comprehensive genomic profiling of MPN patients at Dana-Farber, we have identified key genetic drivers of MPN progression, which we’re studying in the lab with the goal of identifying novel approaches to therapeutically target these high risk molecular genetic events.
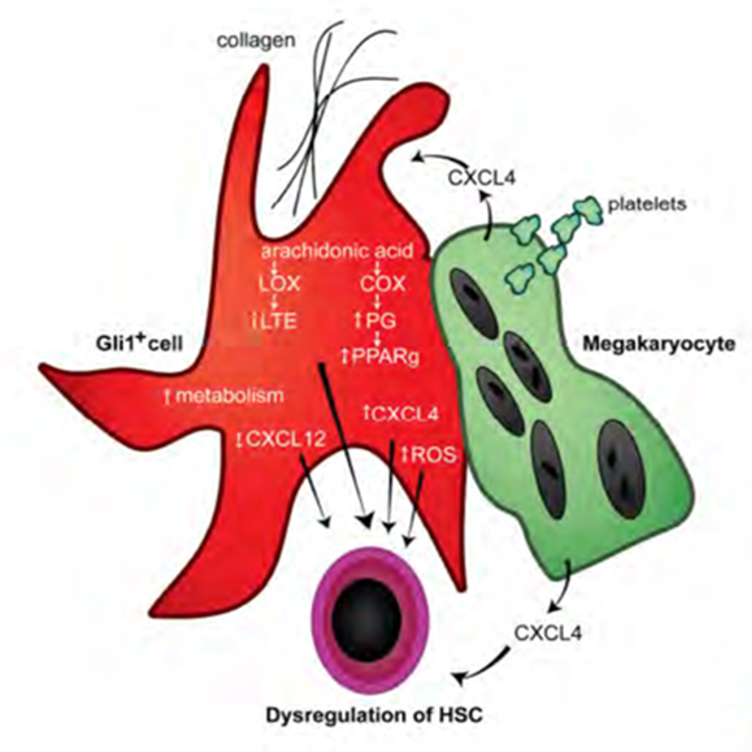
Understanding of the biology underlying myelofibrotic transformation in MPN is quite rudimentary. Our lab is working to understanding the key molecular mechanisms that underlie fibrotic transformation and the key bone marrow cellular populations involved. Using a combination of human and mouse studies and focusing on single cell RNA-sequencing we are working to identify novel biomarkers of myelofibrosis and also have a number of pre-clinical studies testing novel drug treatments with the potential to delay and/or treat myelofibrosis.
We have a longstanding interest in the effects of interferon in MPN, particularly its effects on MPN stem cells. We’re working to identify molecular mediators of resistance to interferon in MPN with the goal of developing new approaches to overcome resistance in patients.